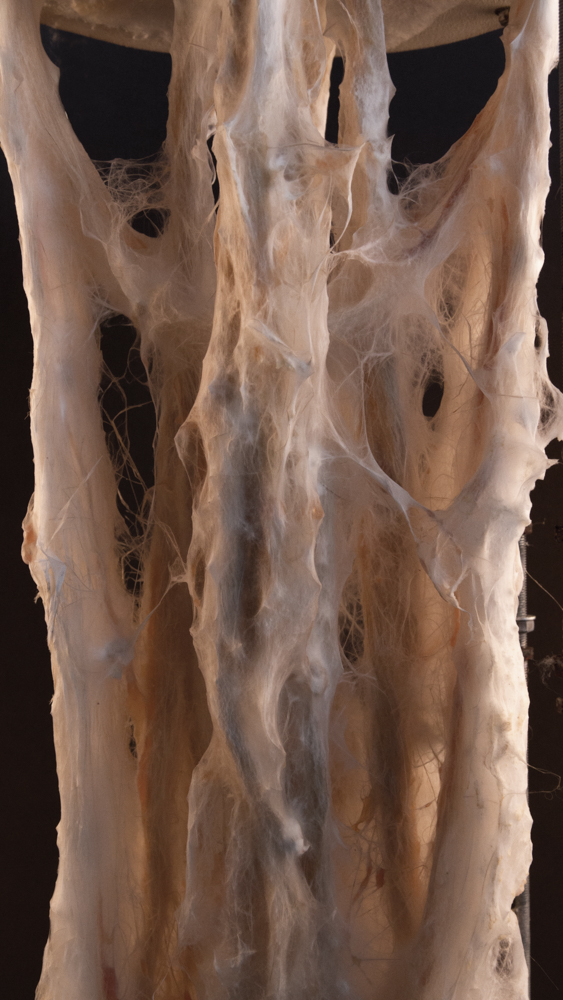
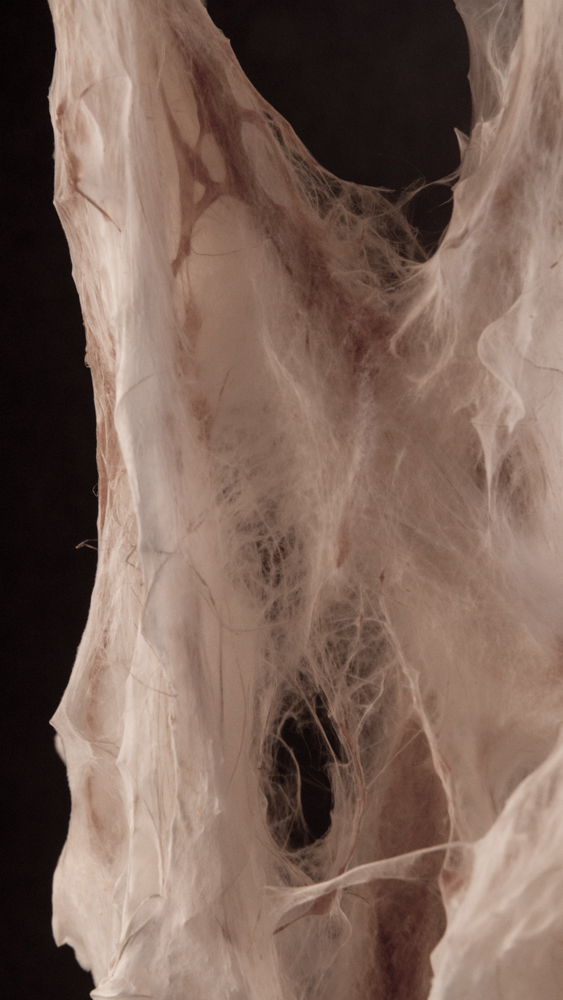
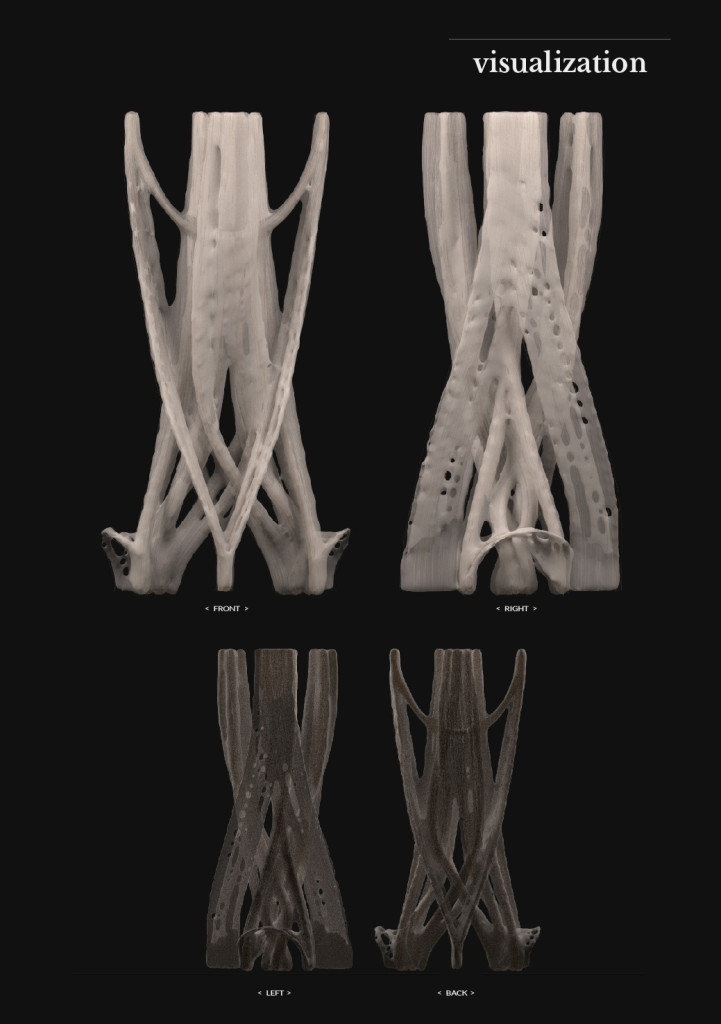
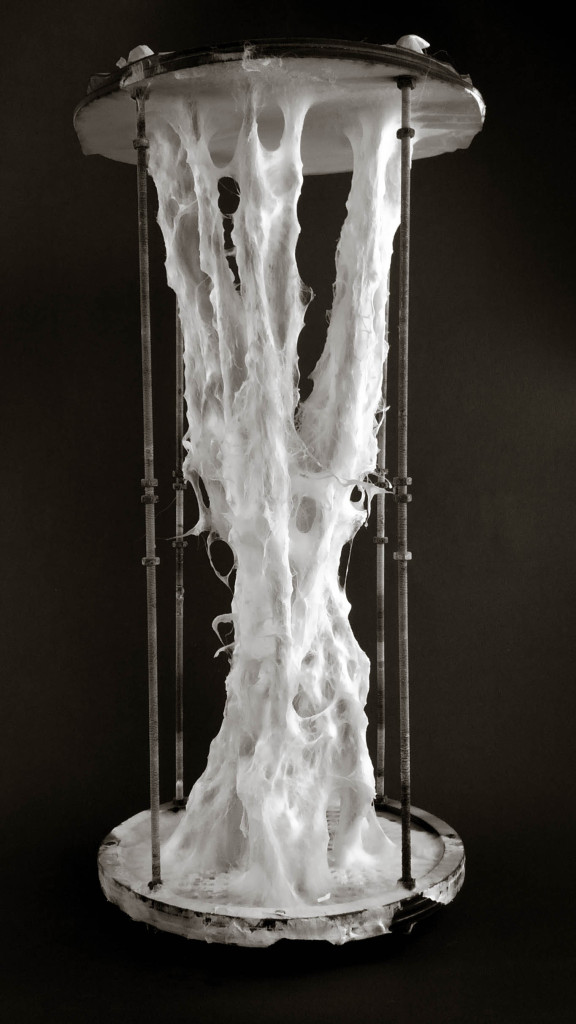
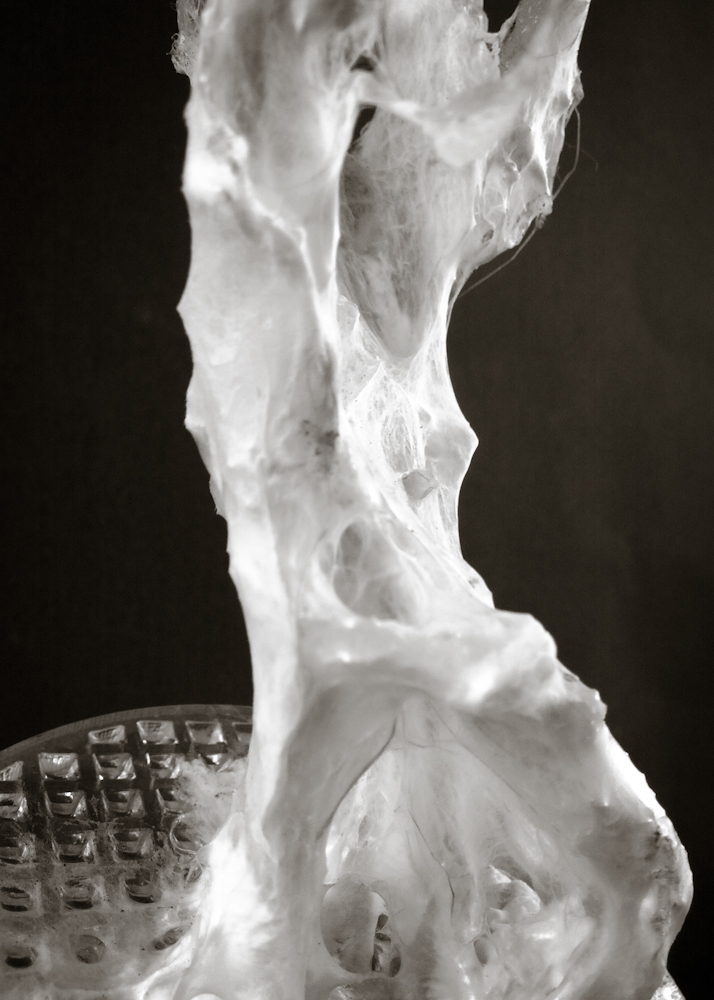
Big model | selfstanding
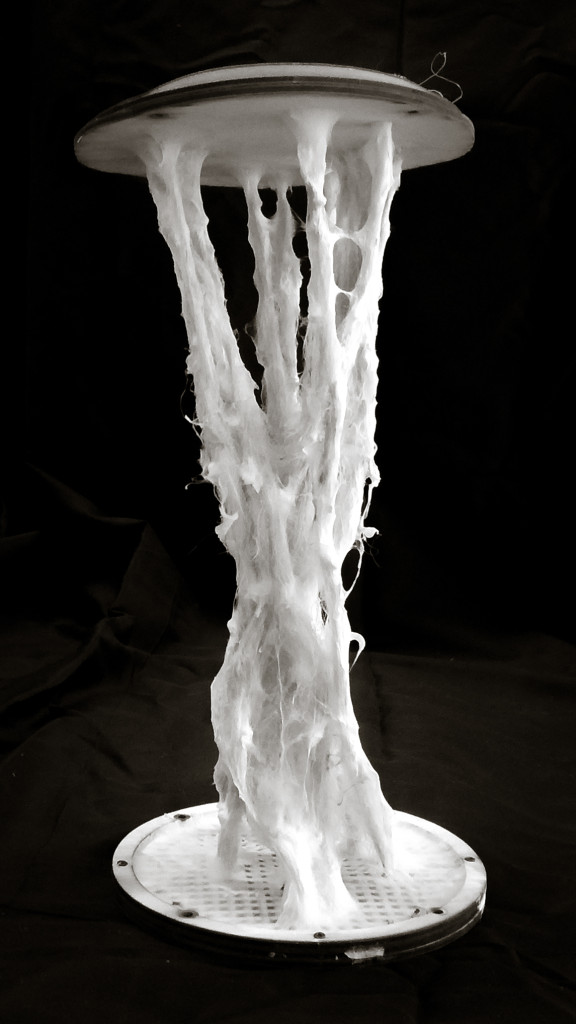
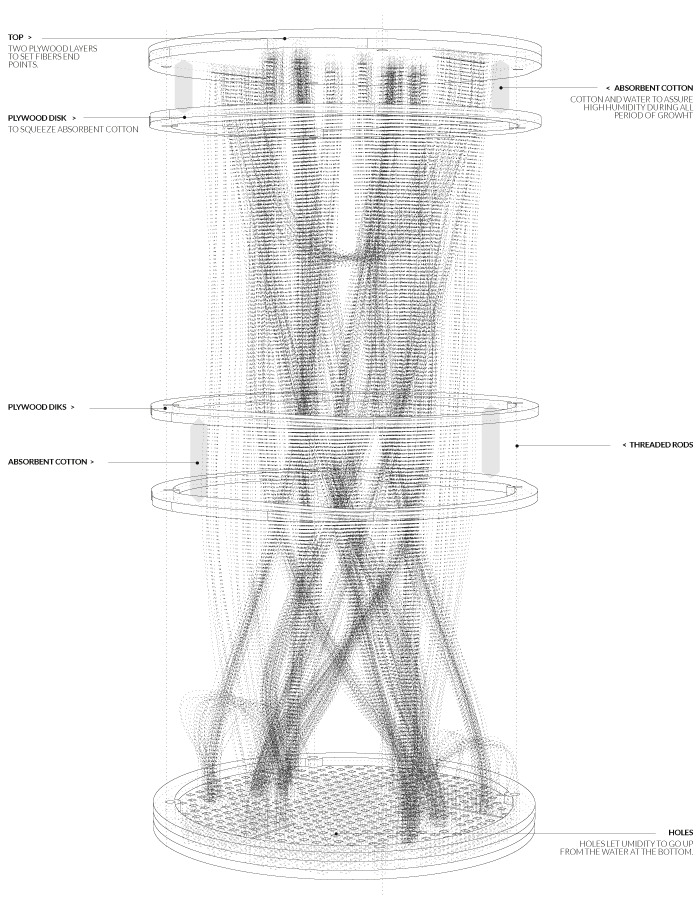
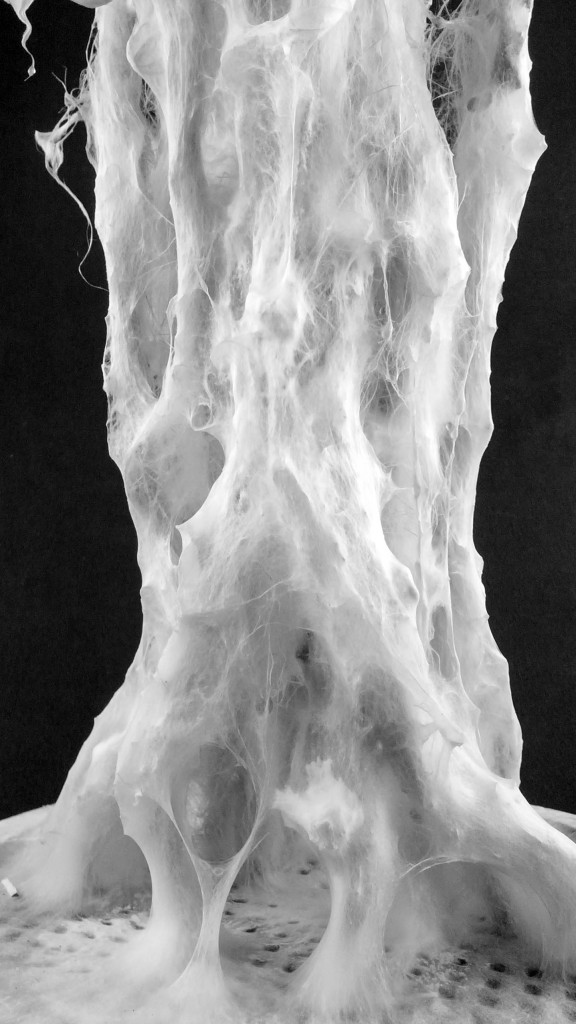
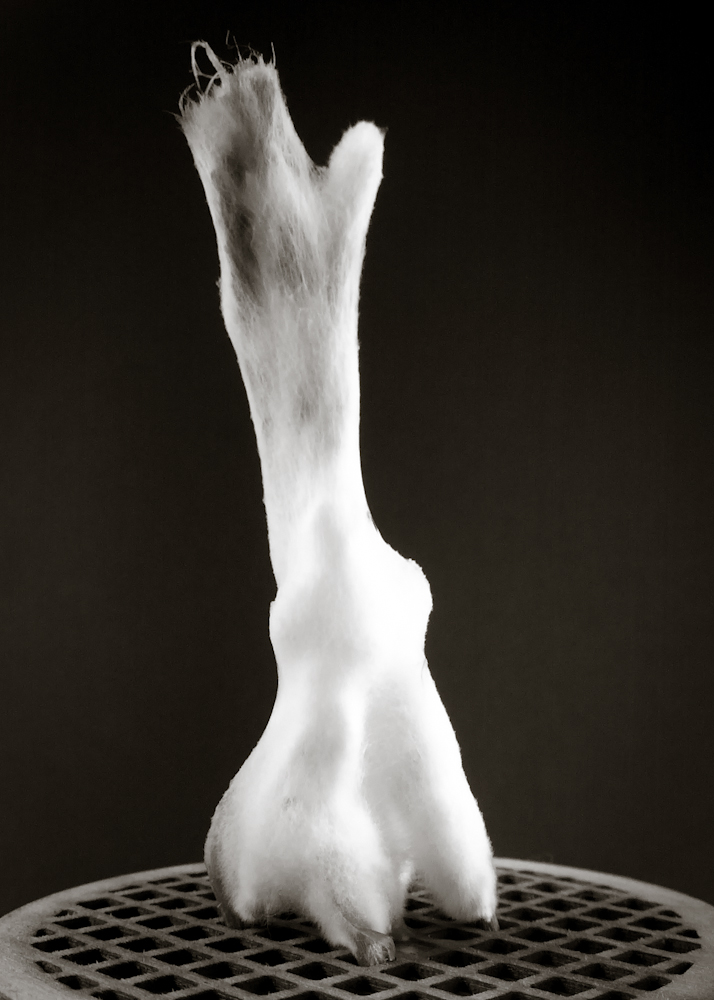
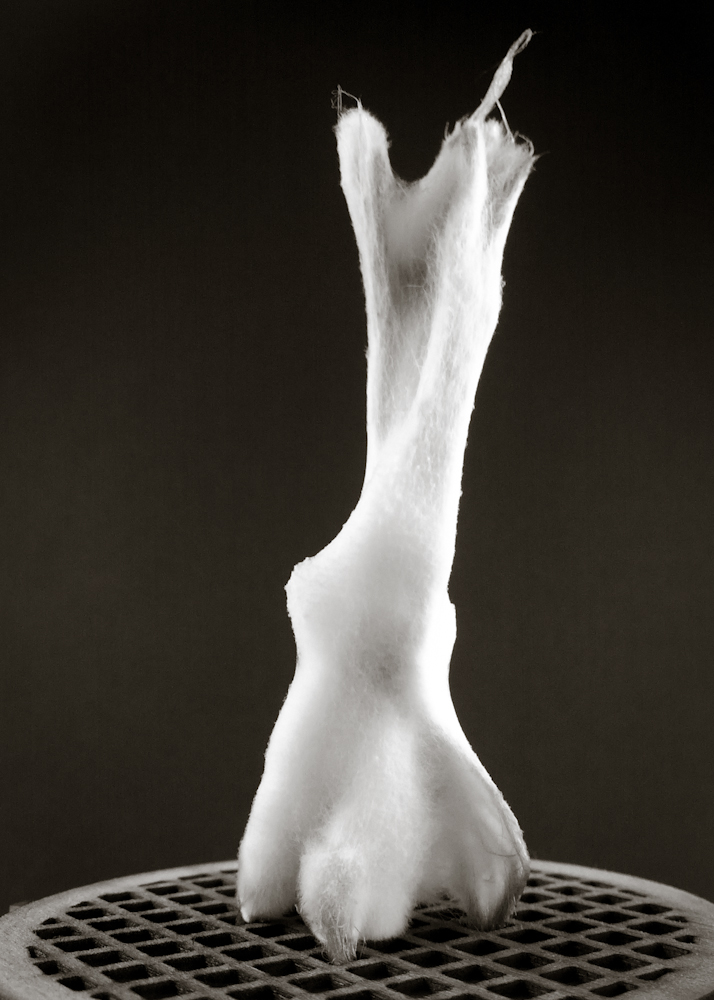
Final model | fabrication
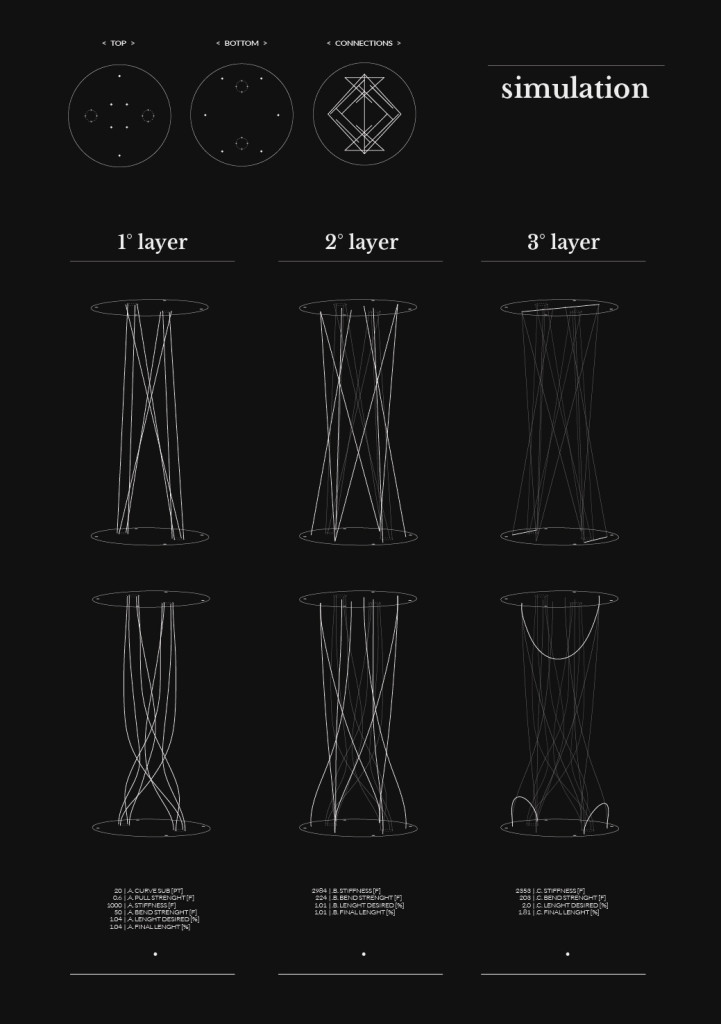
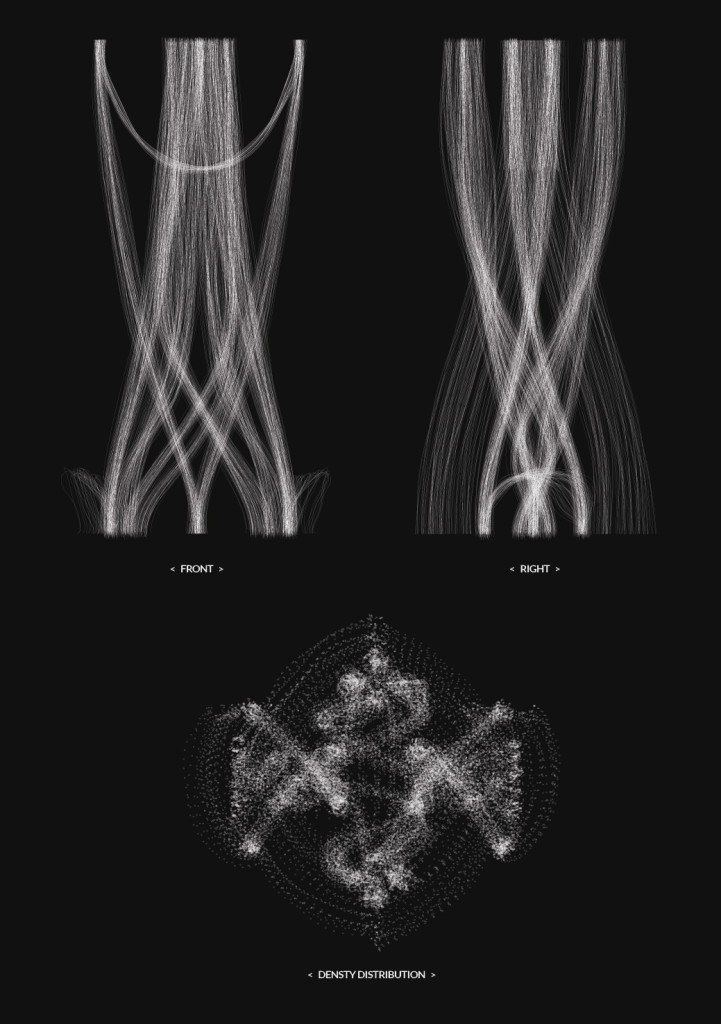
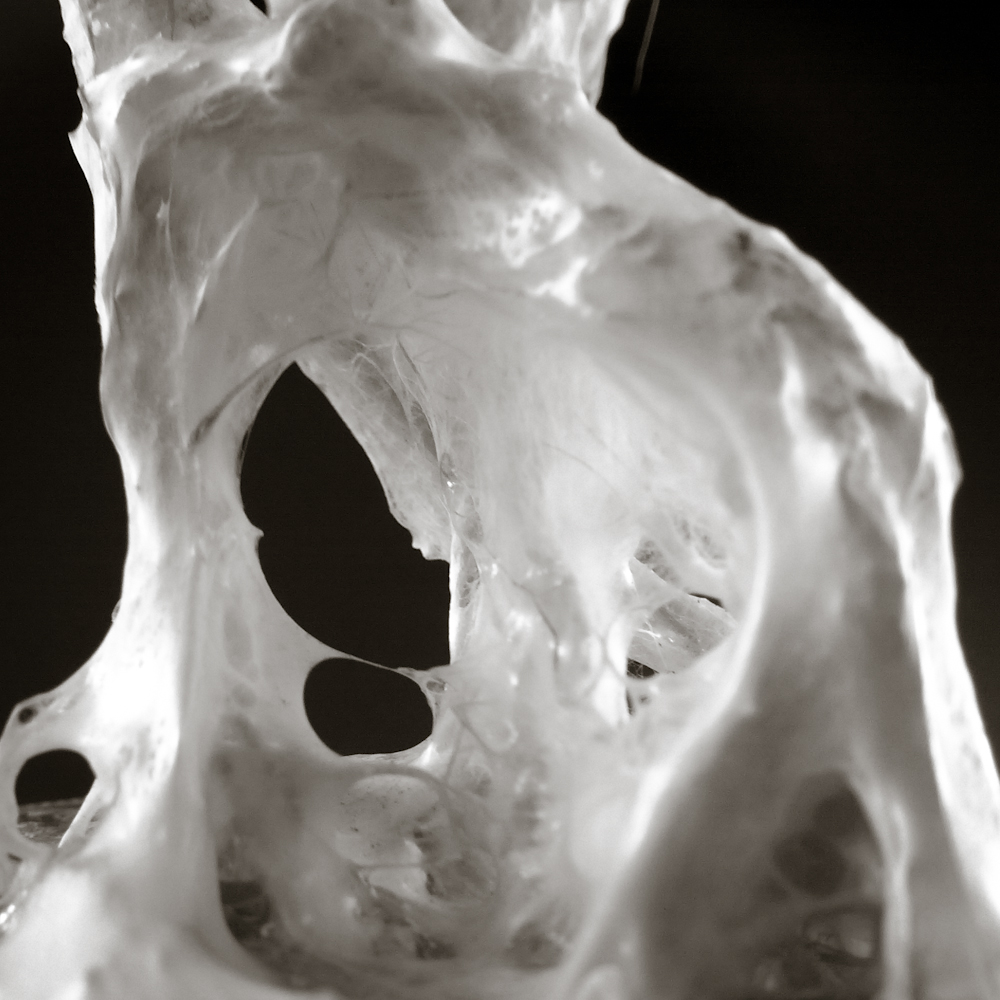
Final model | simulation
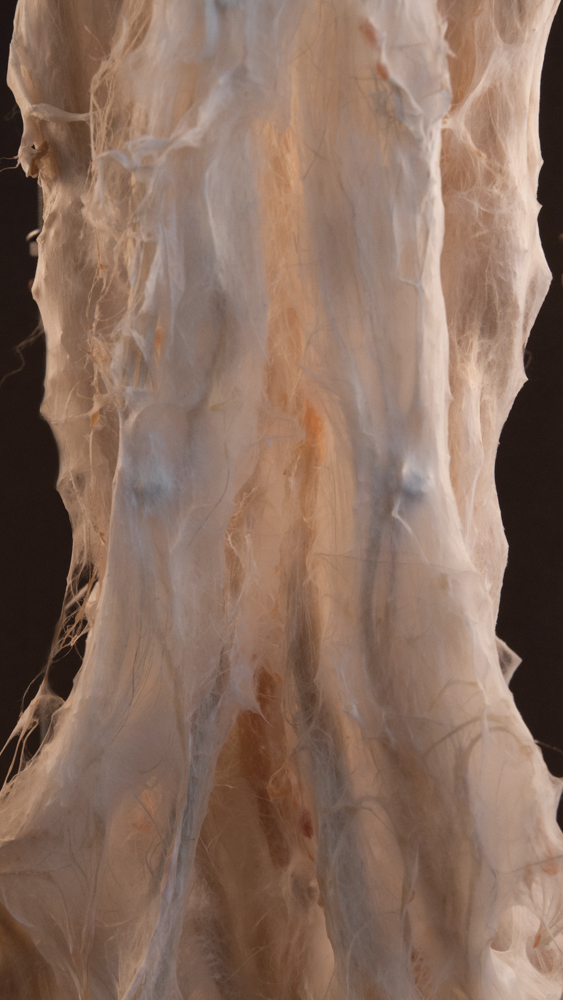
Hemp simulation 02 | explorations
Big model, 350mm high _ 01
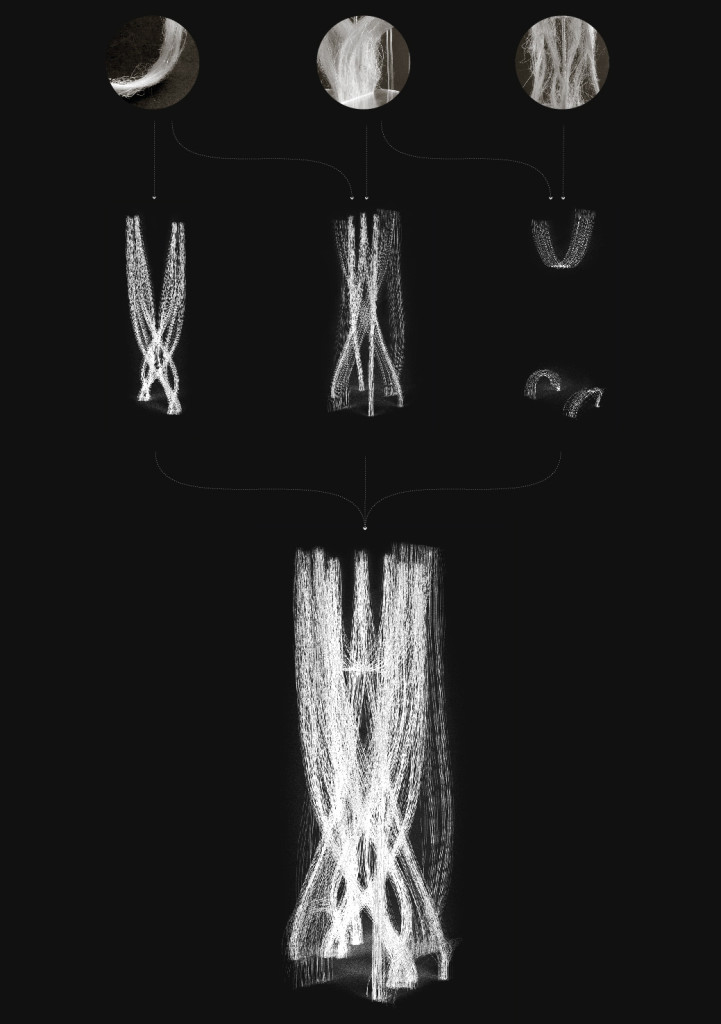
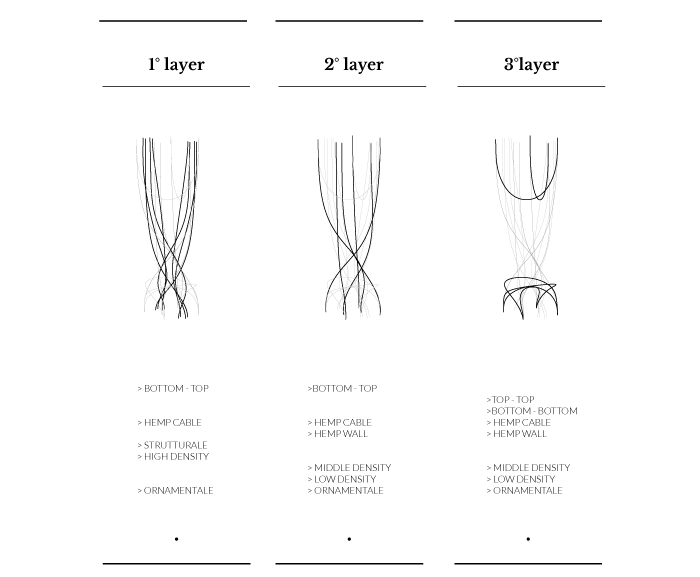
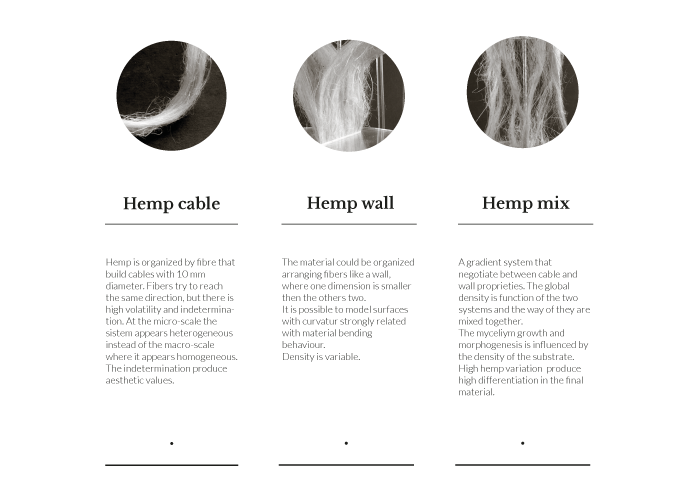
Hemp simulation
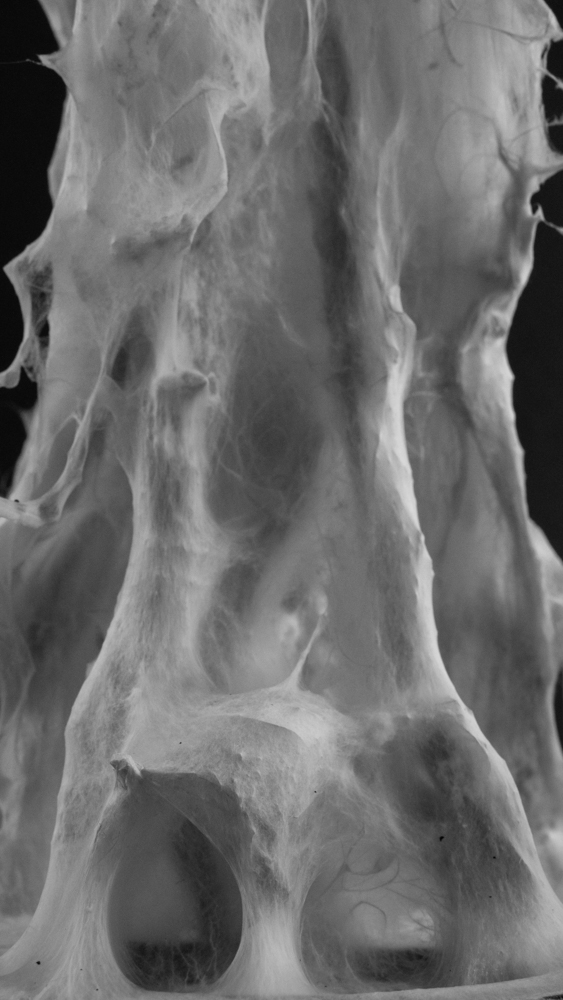
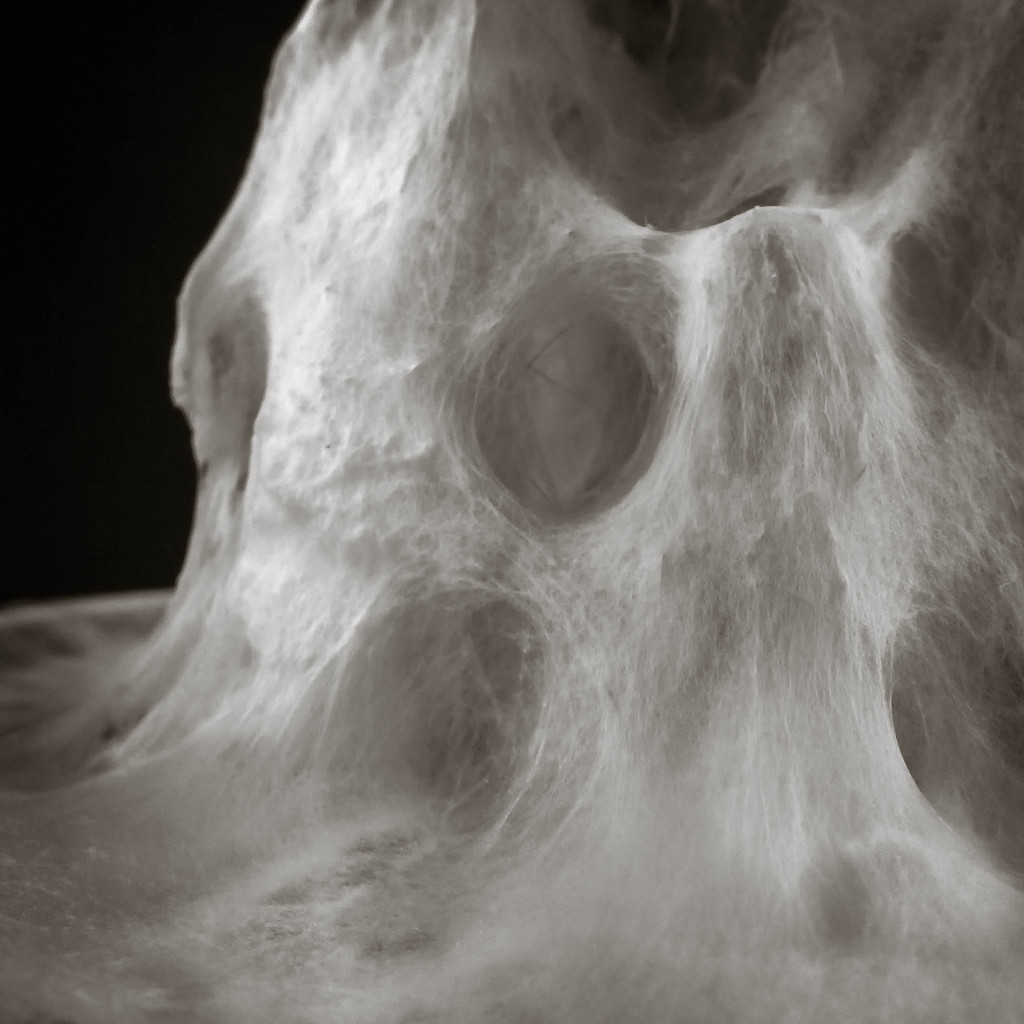
Hemp structure – Gradient
Hemp structure – Walls
Tests aim to explore system behaviours on a different hemp arrangement. Two kinds of “walls” follow the hemp bending.
Sequential operations
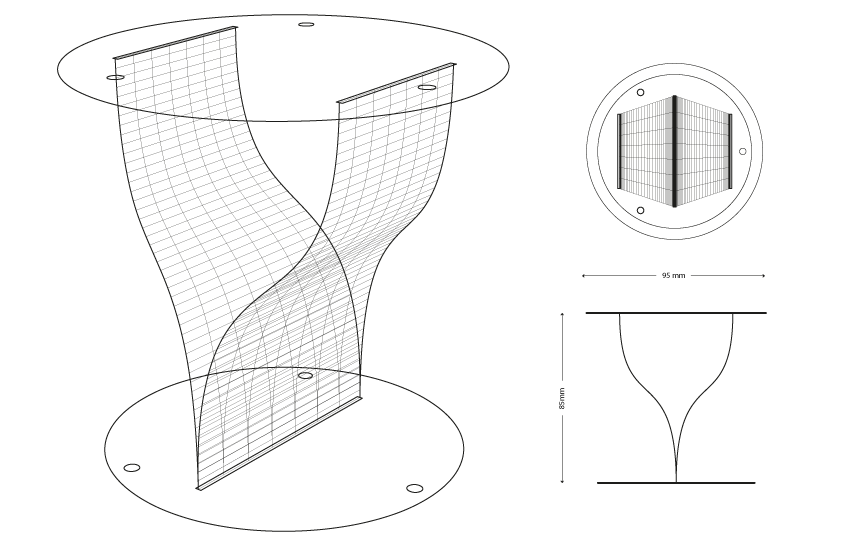
- design and lasercut the plexi structure: plexiglass, diameter 95mm, thickness 2mm (thanks to MakeInBo)
- build hemp structures: distance between plates 85/50mm
- put the structure in a glass jar with some ml of water
- autoclave, 121°C – 20′
- (bio)plastic #2.1.2 spread on fibers
- inoculate with POS1 – Pleurotus Ostreatus
- growth in a dark room at 22.5°C for 18 days
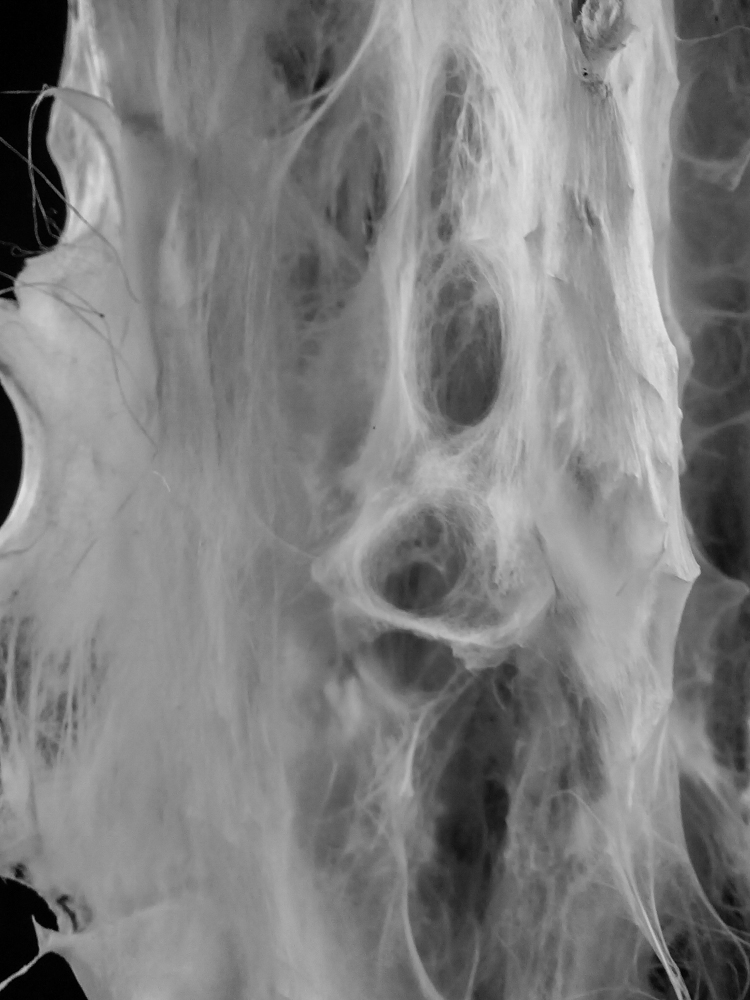
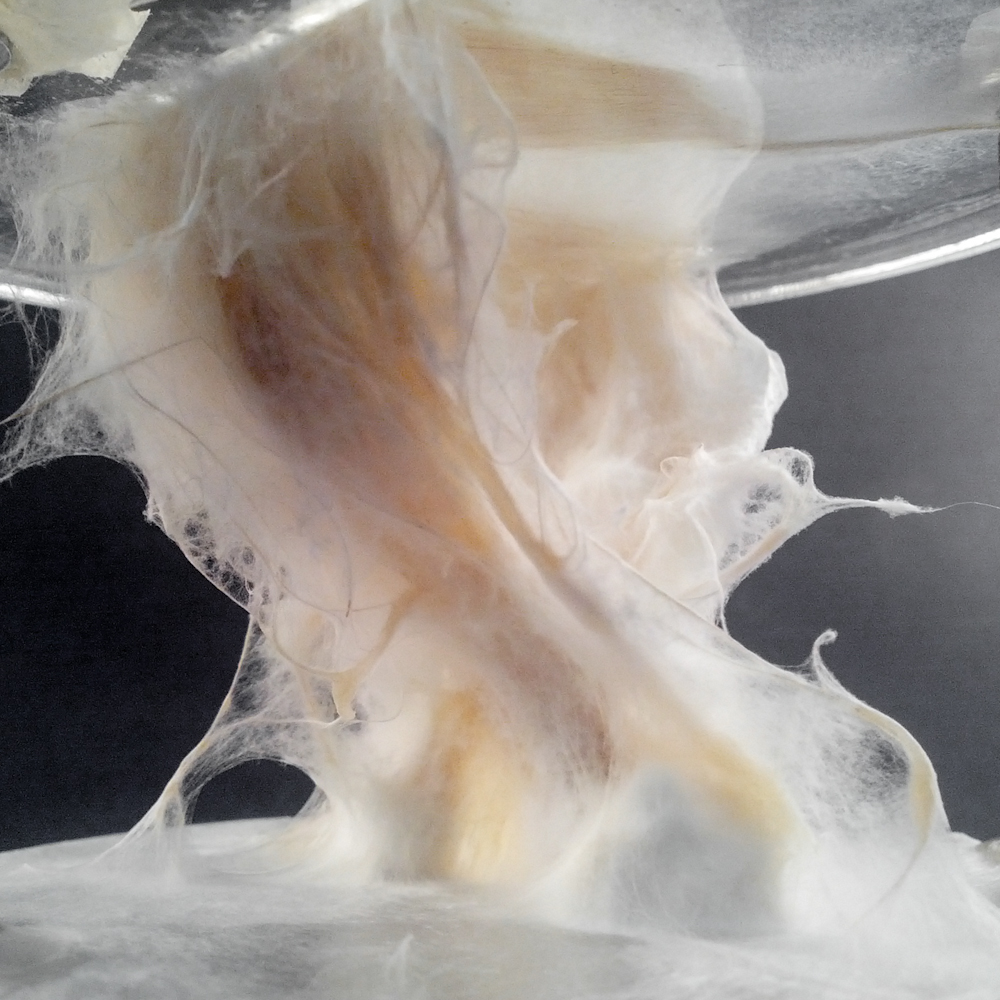
Sample: #3.2.1 _ Simmetric Wall
Sample: #3.2.2 _ aSimmetric Wall
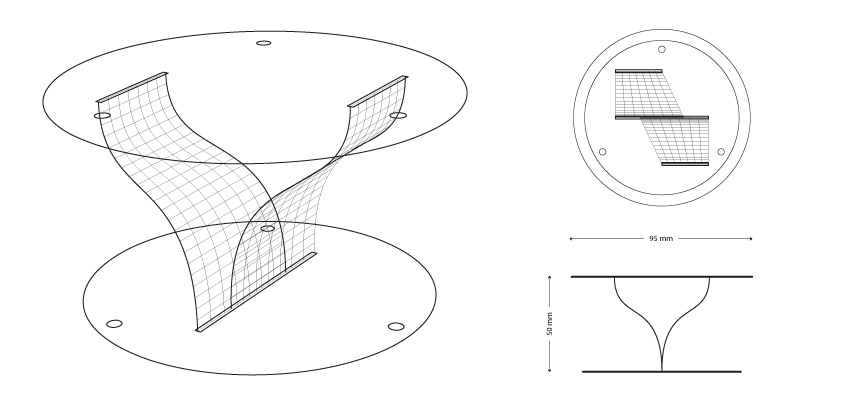
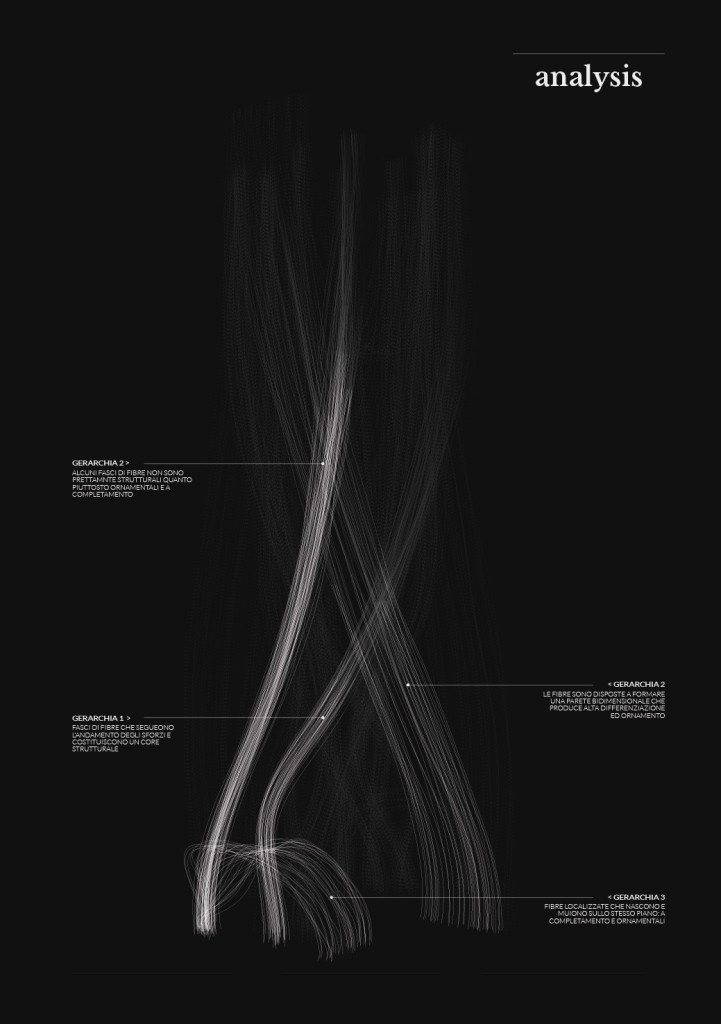
bio-analogic computation
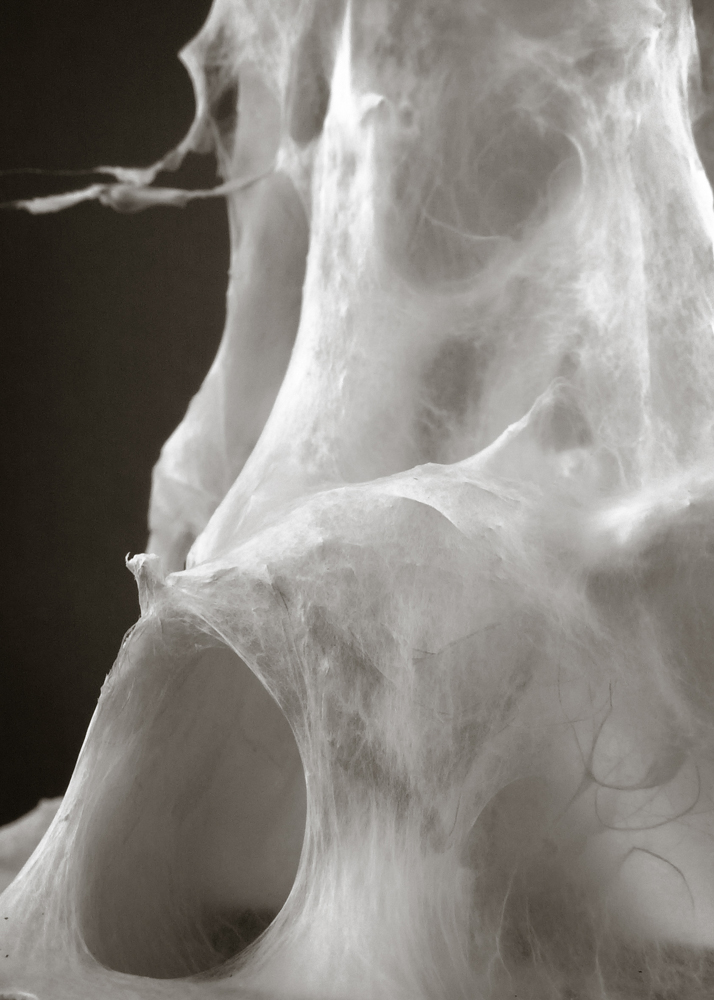
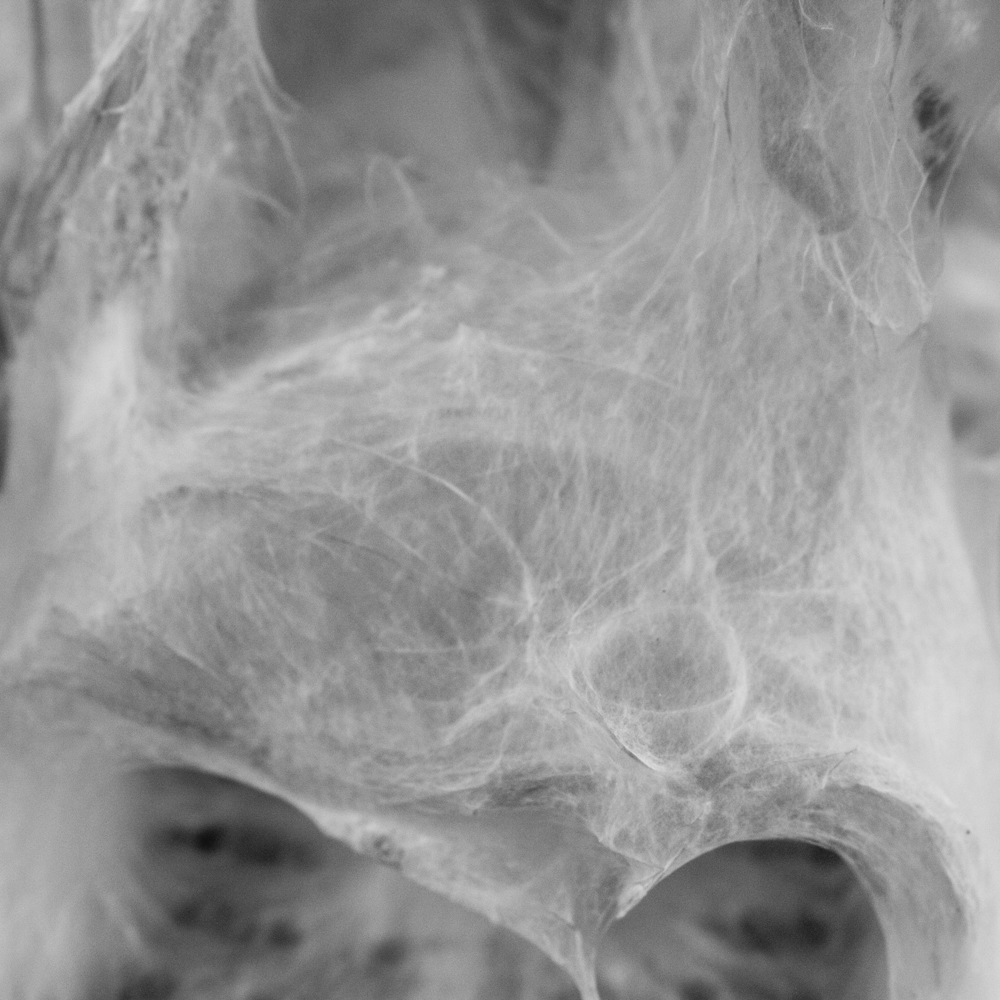
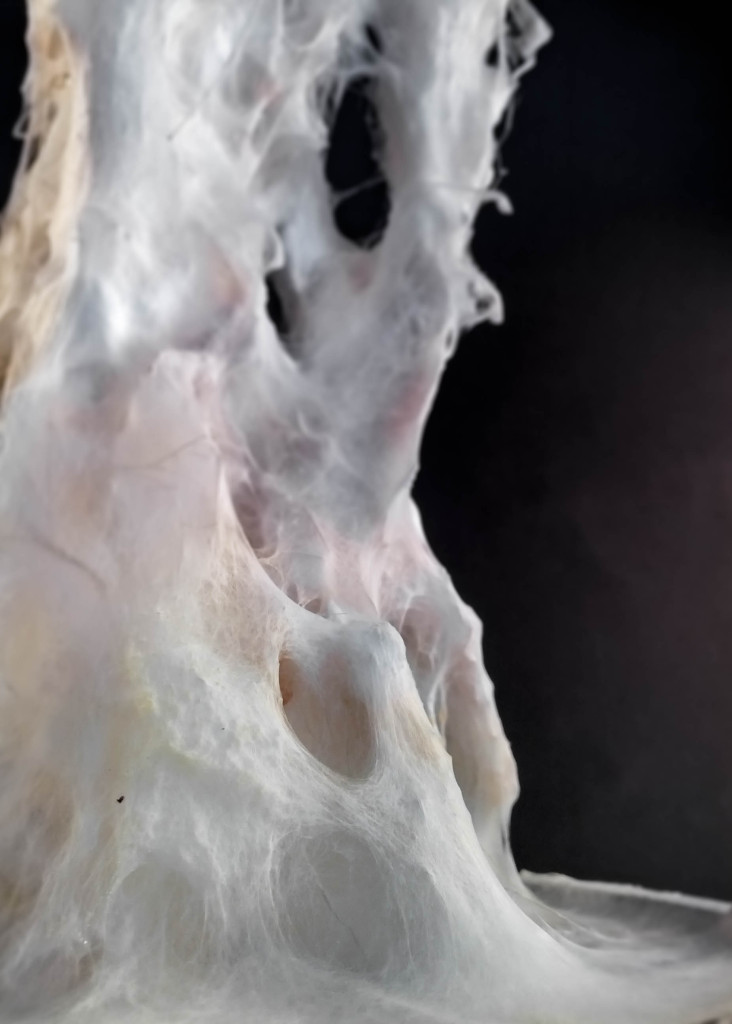
Mycelium is a living agent-system. The hemp fiber structure built here connects two plexiglass plates and It provides a good substrate to mycelium growth. Result shows a redundant and hyper connected system: a fibrous composite material made of (and by) mycelium.
Sequential operations
- design and lasercut plates: plexiglass, diameter 75/88 mm, thickness 3mm (thanks to MakeInBo)
- build hemp structures: distance between plates 15cm
- put the structure in a glass jar with some ml of water
- autoclave, 121°C – 20′
- (bio)plastic #2.1.2 spread on fibers
- inoculate with POS1 – Pleurotus Ostreatus (5 infected wheat grains)
- growth in a dark room at 22.5°C for 12 days > image
Sample #3.1.1 – bio-analogic computation _ mycelium tectonics
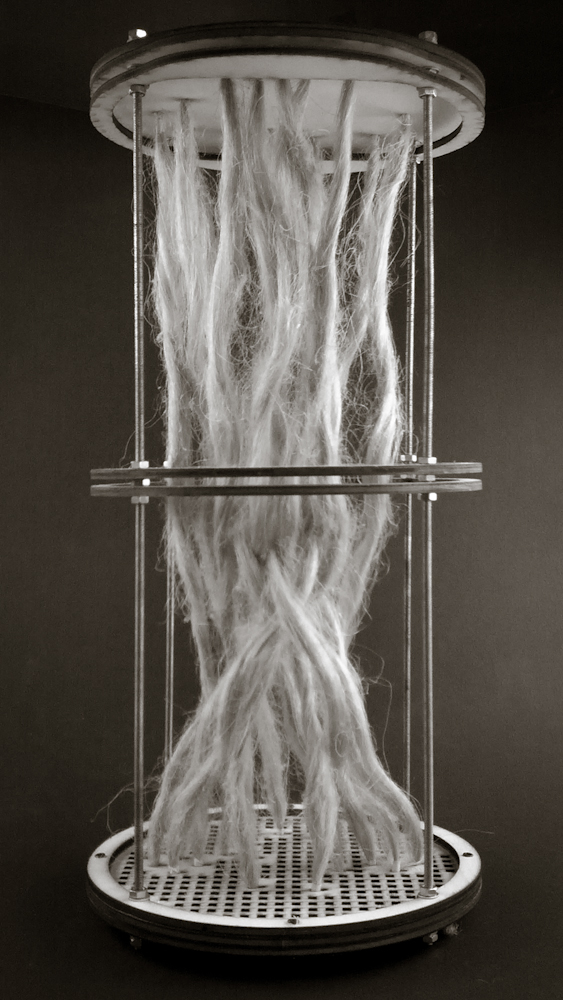
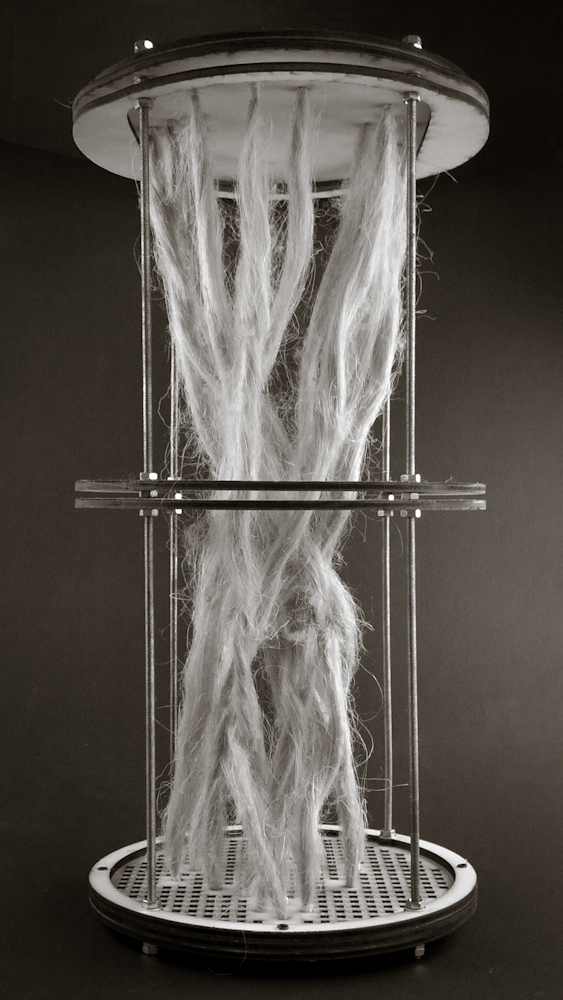
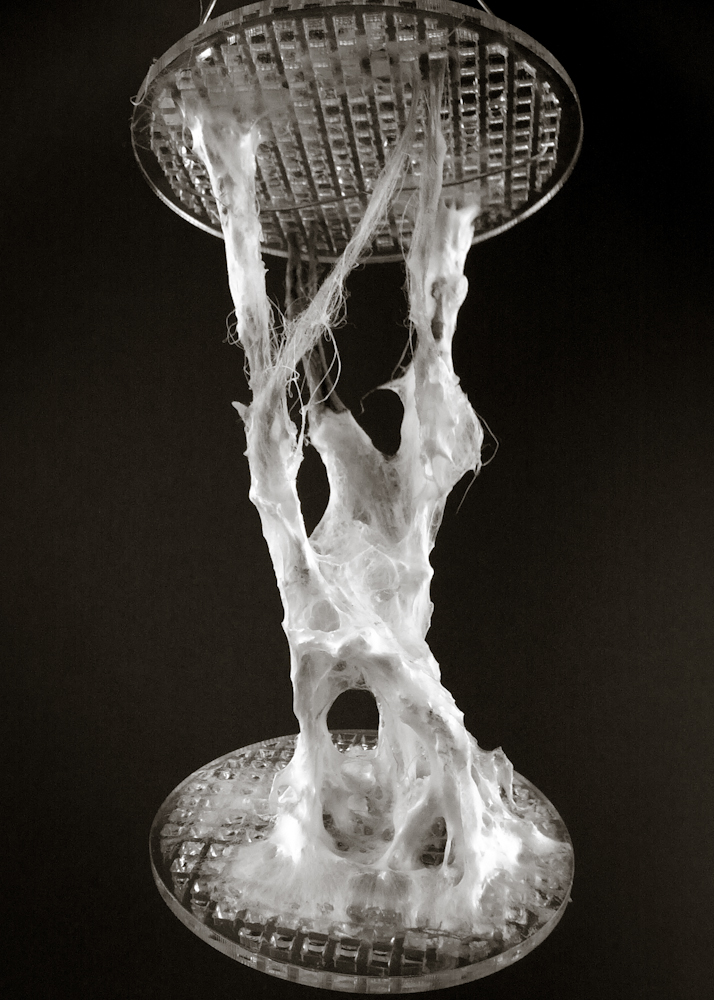
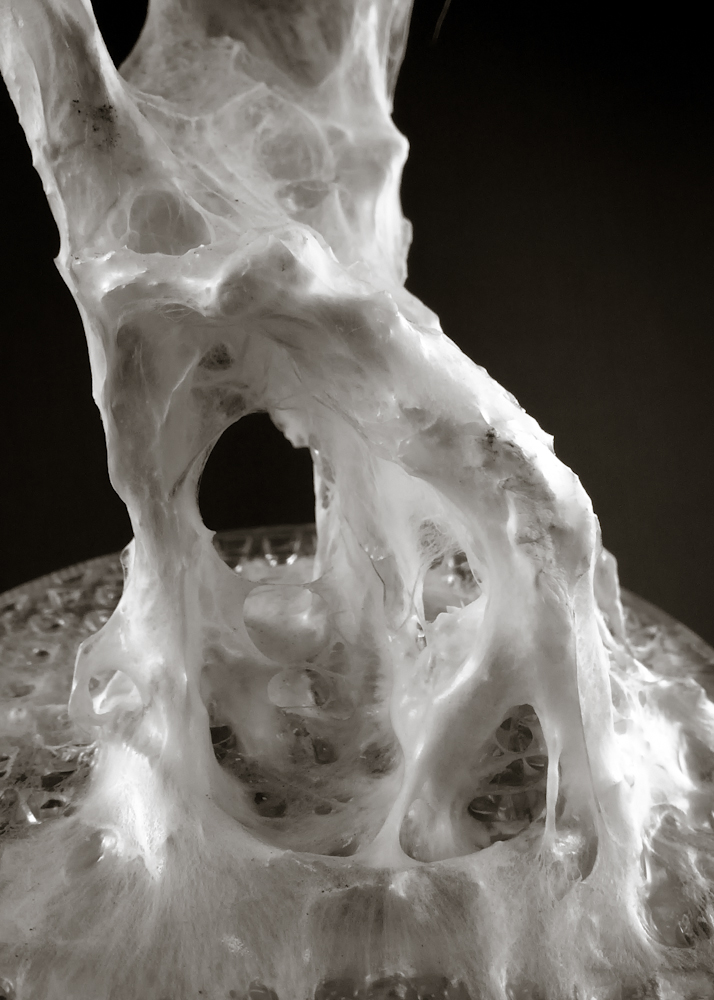
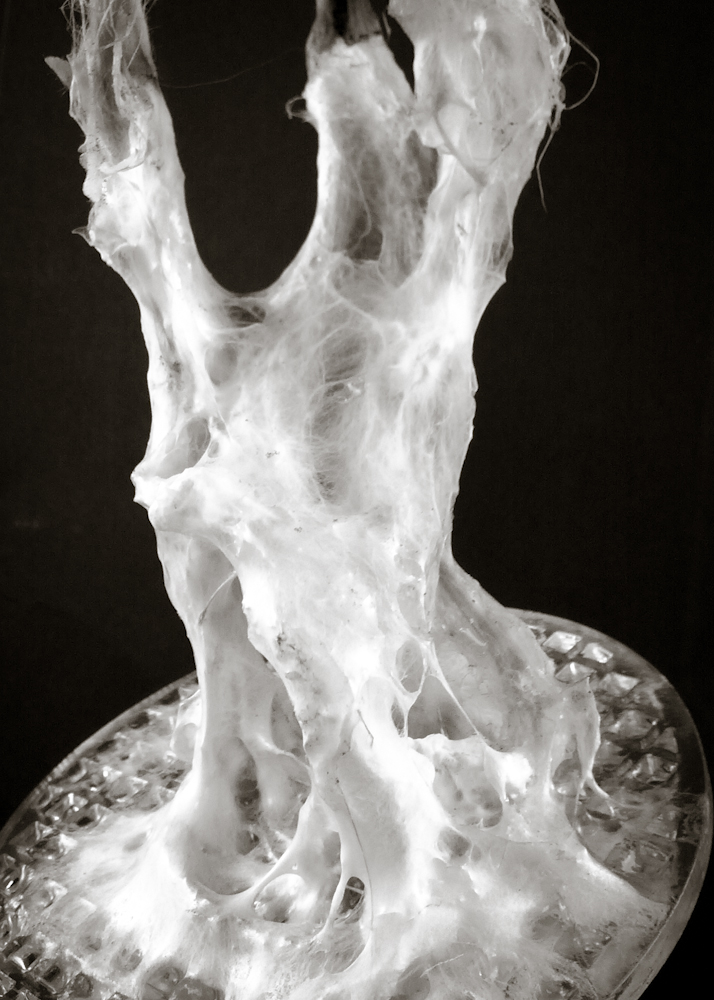
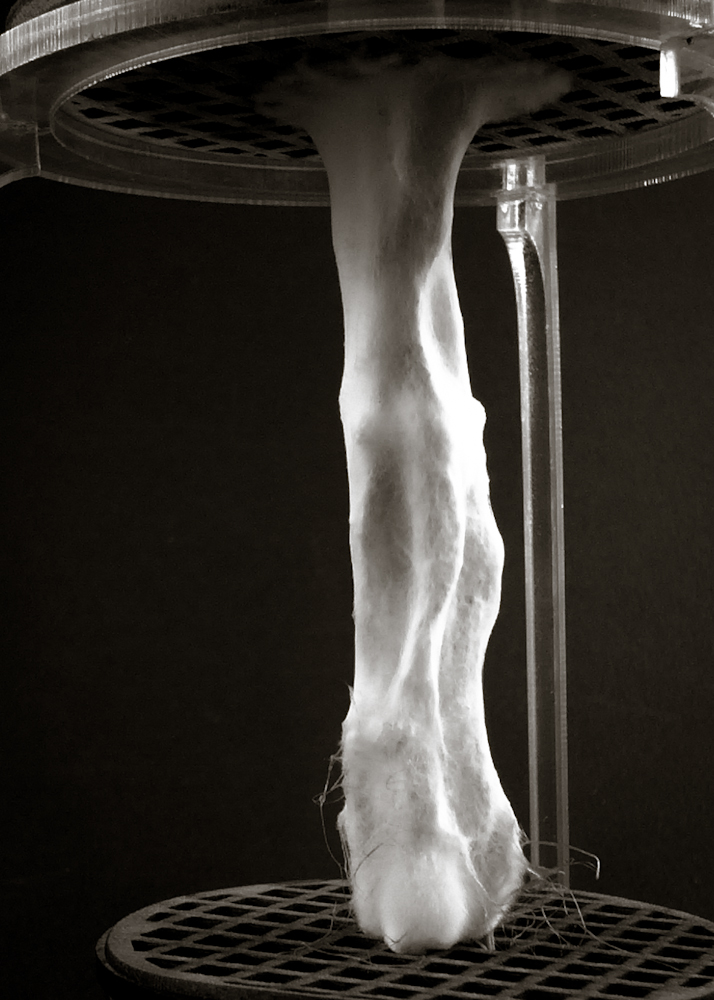
Hemp structure – benchmark
After latest and satisfying inVitro results (1, 2), some simple structures eneble the possibility to investigate the 3th dimension. These “benchmark” are a kind of analogic computation to understend and finally explore mycelium tectonics, architecturally speaking.
Sequential operations
- design and lasercut the wood structure: poplar, diameter 60mm, thickness 4mm (thanks to MakeInBo)
- build hemp structures: distance between plates 10cm
- 24h in water (this step involved fiber bending and volume reduction)
- autoclave, 121°C – 20′
- (bio)plastic #2.1.2 spread on fibers > image
- inoculate with POS1 – Pleurotus Ostreatus (one infected wheat grain)
- put the structure in a glass jar with some ml of water to ensure humidity > image
- growth in a dark room at 22.5°C for 16 days > image
#3.0.1B – “gravity”
#3.0.2B – “double plate linear”
#3.0.3B – “double plate open”
#3.0.4B – “double plate bend&torsion”
—
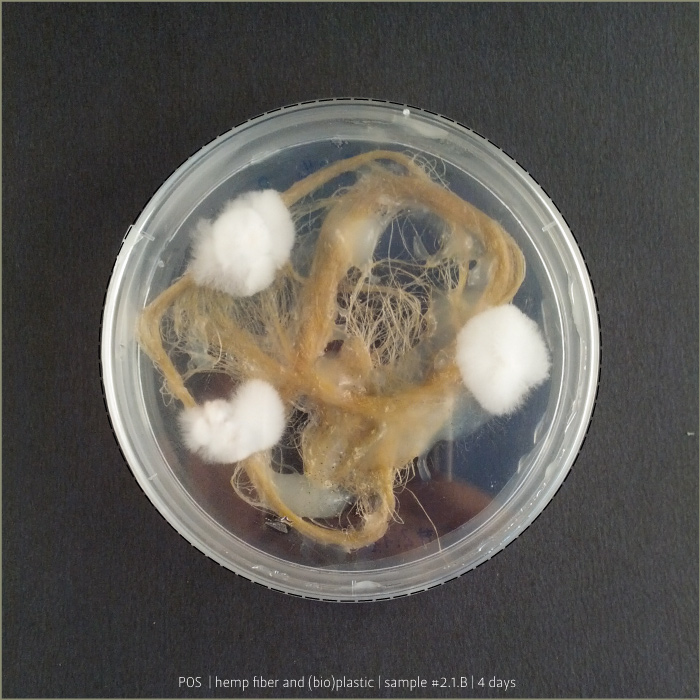
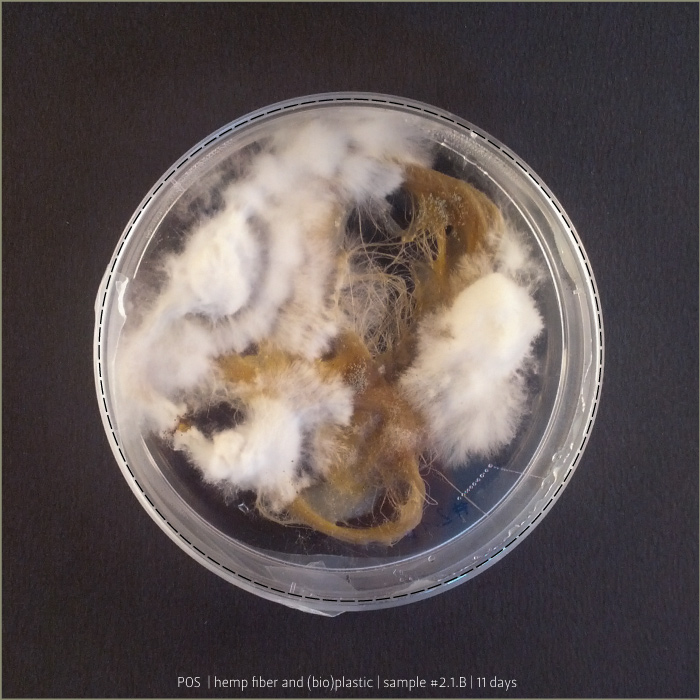
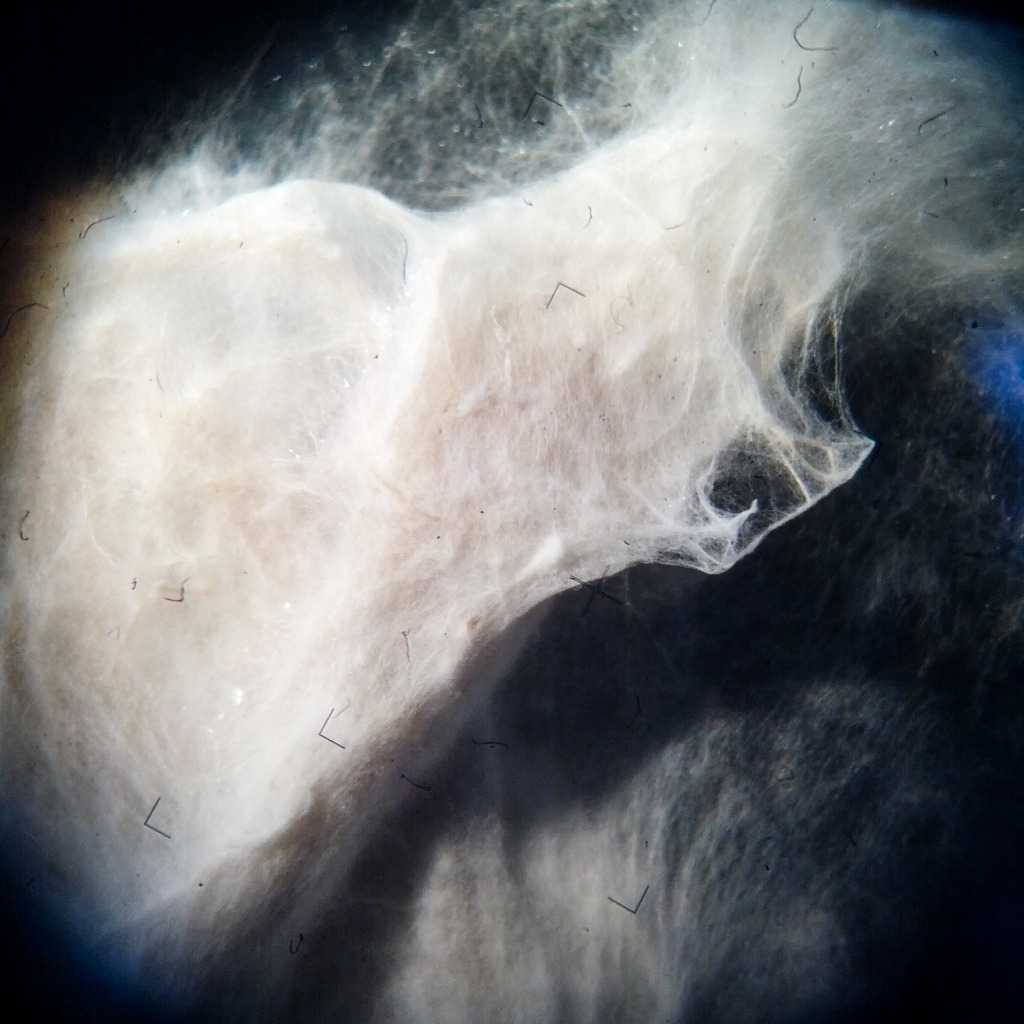
Pleurotus & Fiber & (bio)Plastic
A test to explore mycelium behaviours on a substrate made by hemp fiber and (bio)Plastic. Once Pleurotus is sufficiently grown, petri capsula was opened to let it air-dry.
Results show an high tensile strength fibrous composite.
(bio)plastic recipe: #2.1
mycelium: POS 1 _ Pleurotus Ostreatus
Achim Menges 01
“Natural morphogenesis, the process of growth and evolutionary development, generates systems that derive complex articulation, specific gestalt and performative capacity through the interaction of system-intrinsic material characteristics, as well as external stiumli of enviromental forces and influences. Thus, formation and materialisation are always inherently and inseparably related in natural morphogenesis.”
– Achim Menges
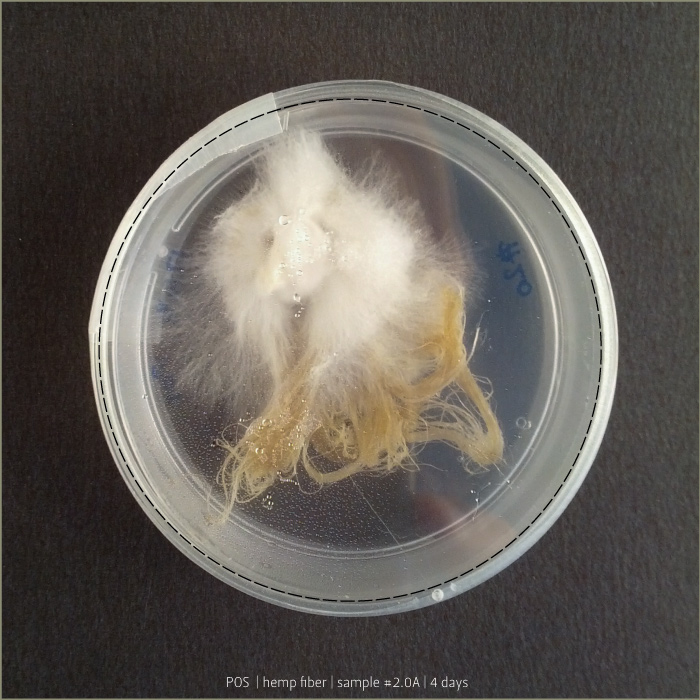
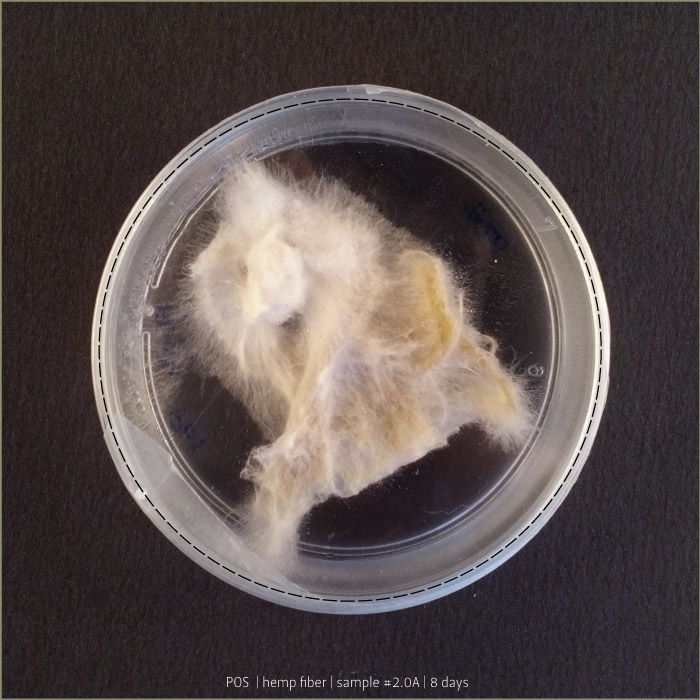
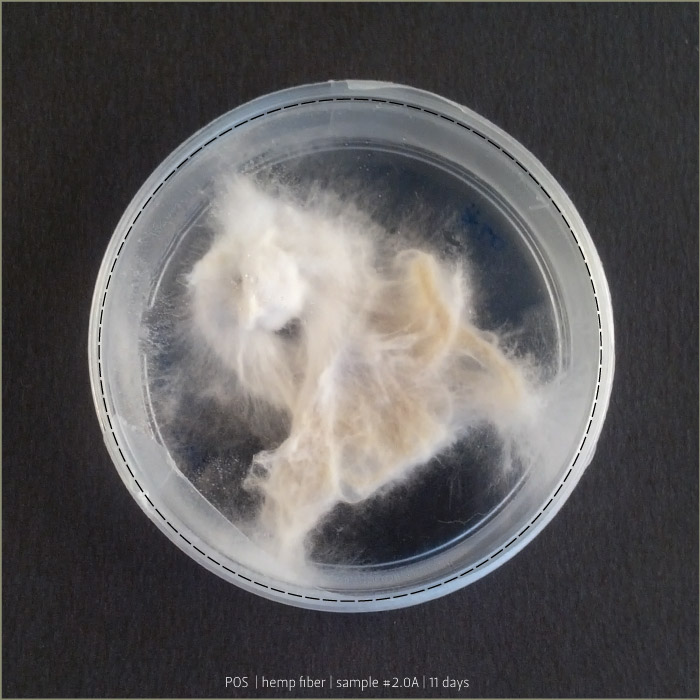
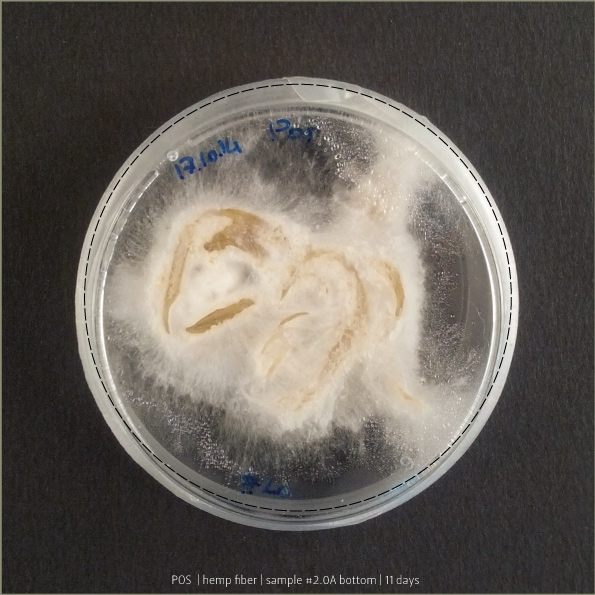
Pleurotus & Fiber
(bio)plastic benchmark
These tests aim to understand if starch plastic could be a good substrate for micelia. After a lot of experiments, finally I found the plastic that has the right consistency to be spread on fiber.
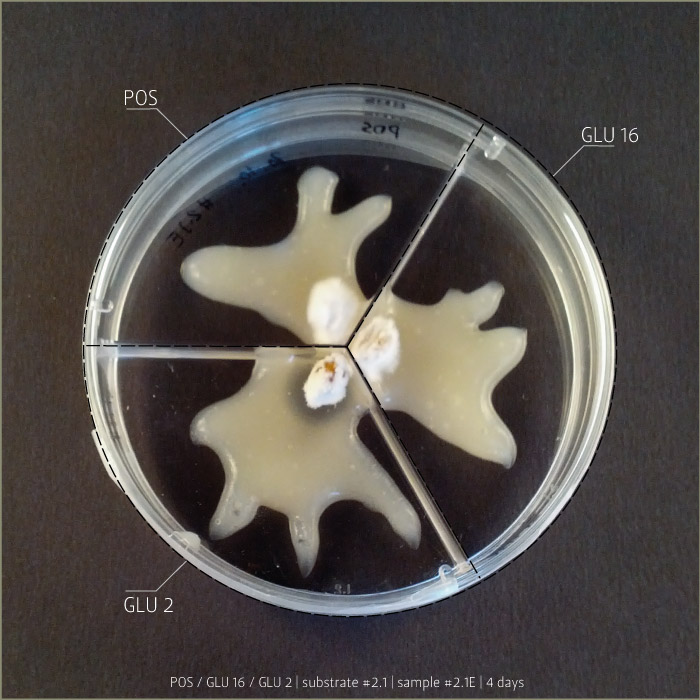
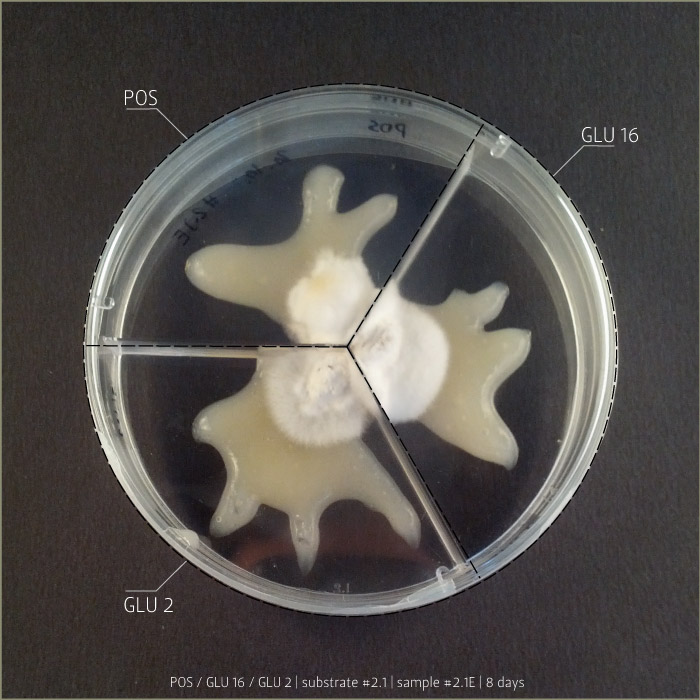
(bio)plastic recipe: #2.1
85% H20
6% Starch
4% Carboxymethyl cellulose sodium salt
3% Glycerol
2% PDA – potato dextrose agar
0.1% Ammonium Acetate
Micelia used for the test:
POS 1 _ pleurotus ostreatus
GLU 2 _ ganoderma lucidum
GLU 16 _ ganoderma lucidum
Recipe #2.1 after 4 days and 8 days:
in vitro petriMask
Exploring boundaries
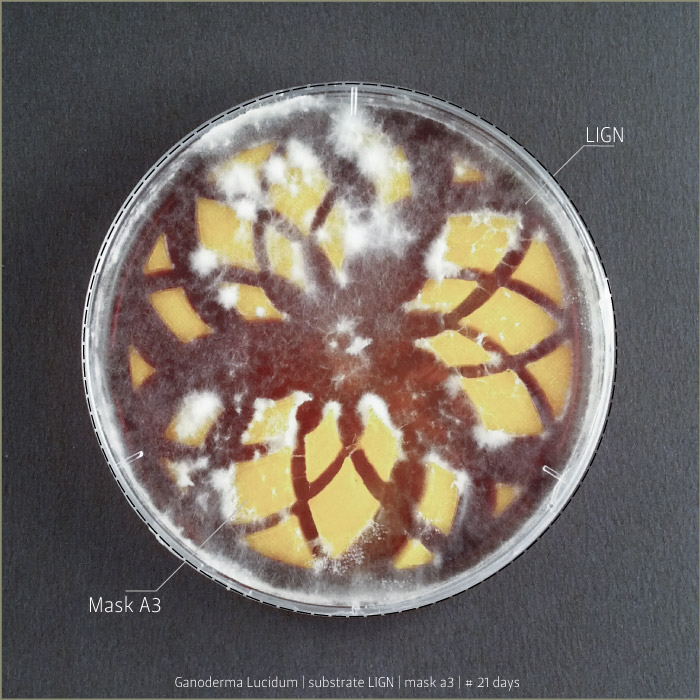
How does the mycelium explore boundaries and build bridge connetions from landscape morphology?
This test allows to understand mycelium’s behaviours of growth through PLA substrate.
To do that a 3d printed mask was insert into a petri dish before the sterilization.
Substrates are:
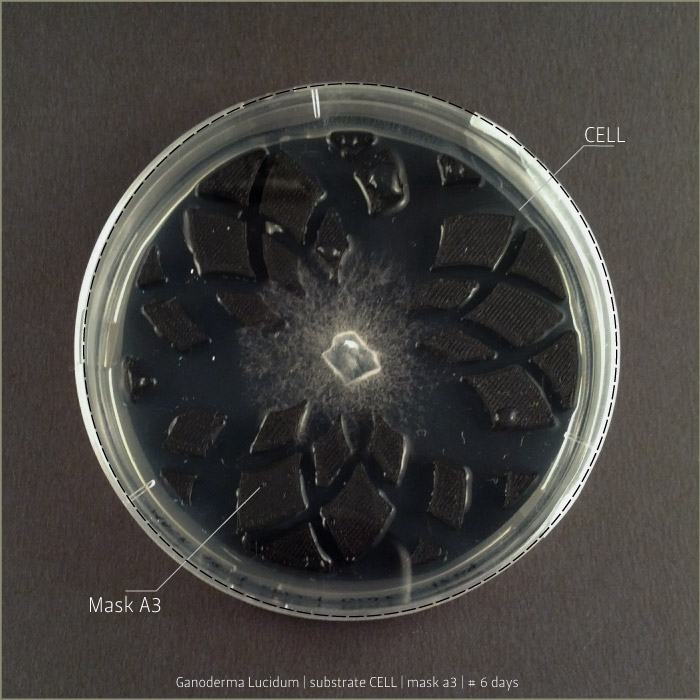
LIGN _ Lignosulphonate bretax C 1,5% + AA 1% | pH: 6,12
CELL _ carboxymethyl cellulose sodium salt 1,5% + AA 1% | pH: 5,92
Micelia are:
GLU2 _ ganoderma lucidum (inoculated on 18th of September 2014)
POS1 _ pleurotus ostreatus (inoculated on 18th of September 2014)
ARM _ armillaria mellea (inoculated on 18th of September 2014)
Thanks to MakeInBo

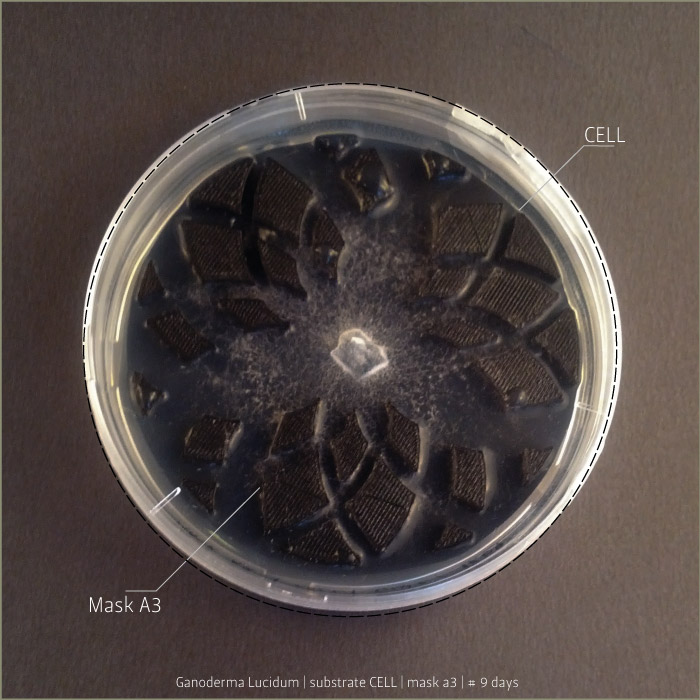
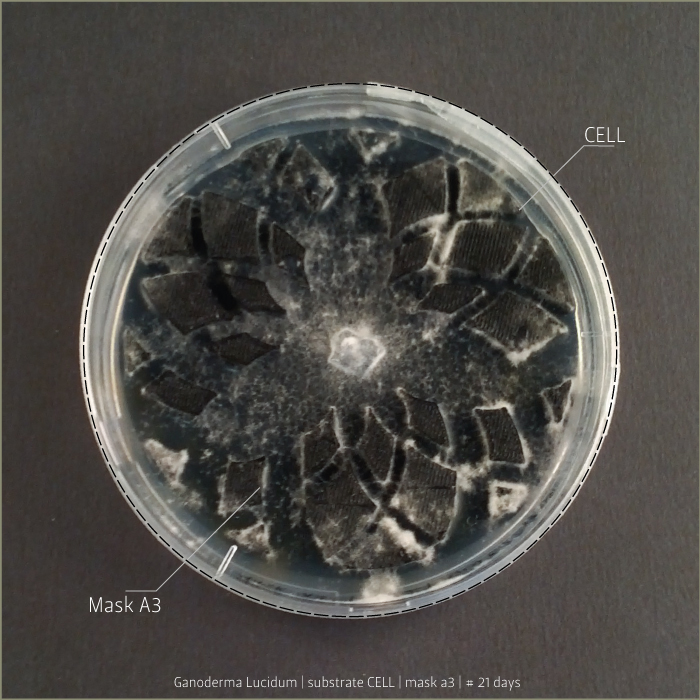
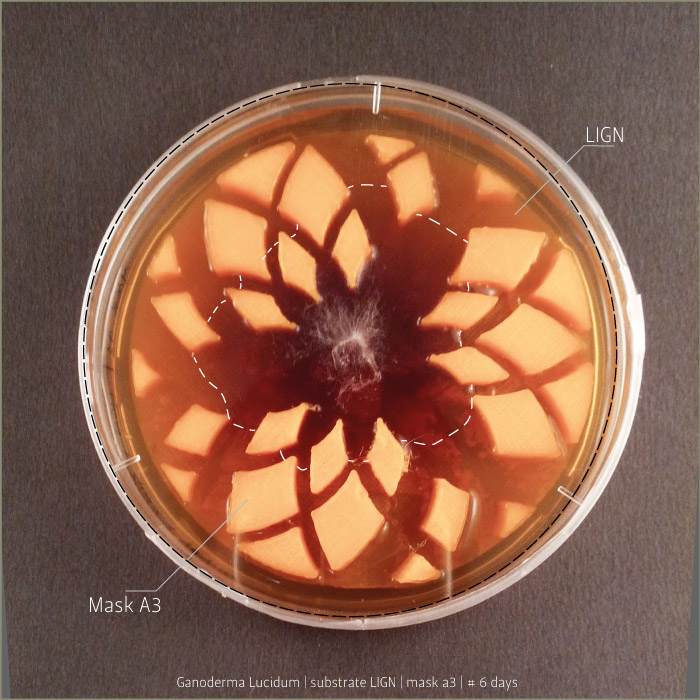
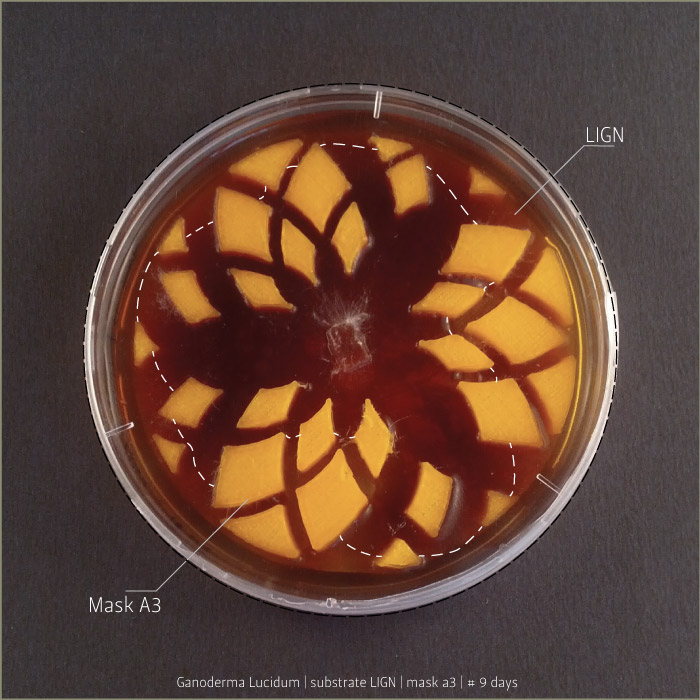
Ganoderma Lucidum after 6 days, 9 days and 21 days:
in vitro mycoTests 02
Previous in vitro mycoTests here.
Lignin and cellulose substrate
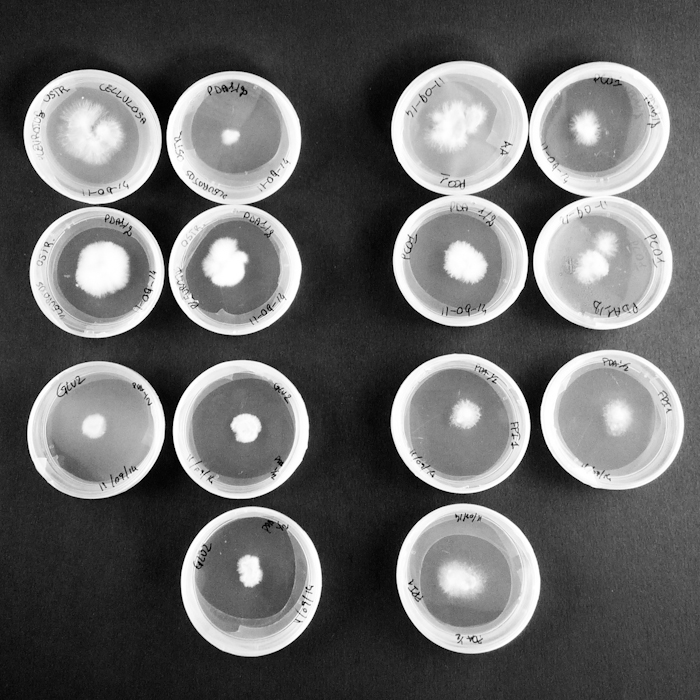
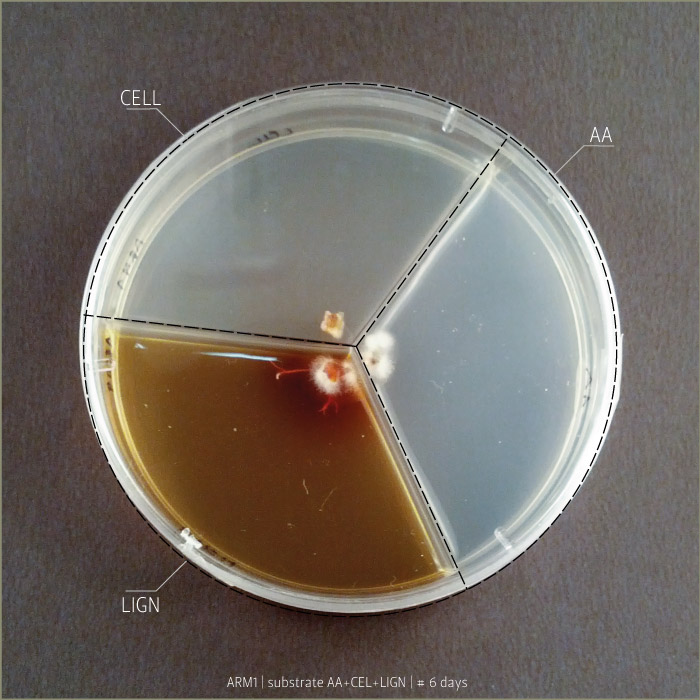
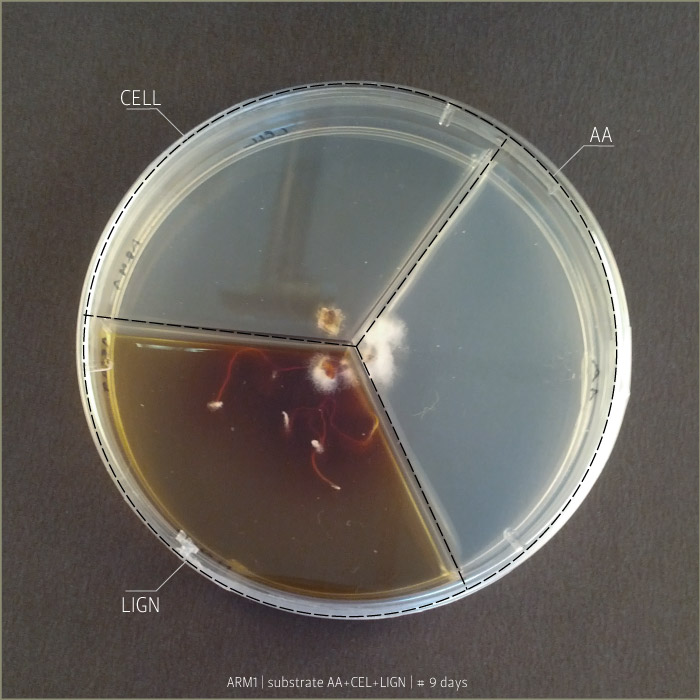
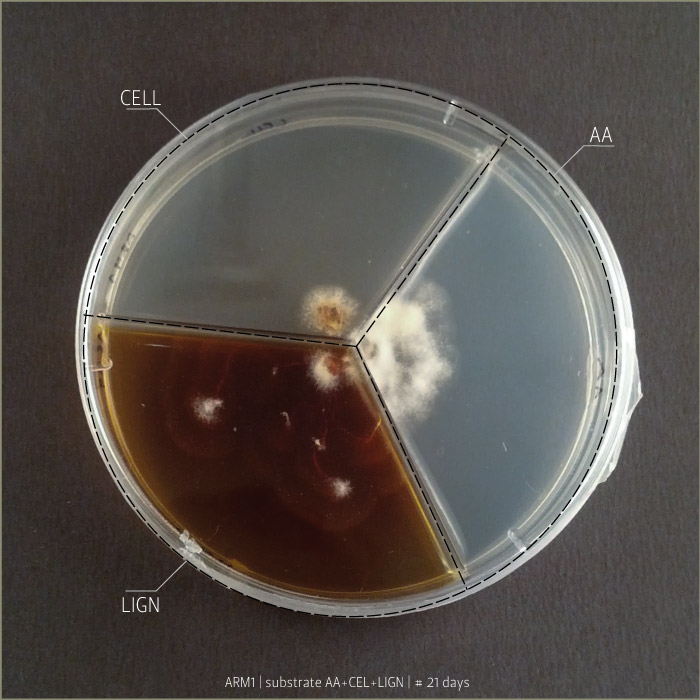
Some tests highlight behavioral growth differences of three species of micelia. Substrates were prepared and filled in petri capsules divided in three sub-compartments.
Substrates are:
AA _ agar agar grade A | pH: 6,038 (test)
LIGN _ Lignosulphonate bretax C 1,5% + AA 1% | pH: 6,12
CELL _ carboxymethyl cellulose sodium salt 1,5% + AA 1% | pH: 5,92
Micelia are:
ARM _ armillaria mellea (inoculated on 18th of September 2014)
GLU2 _ ganoderma lucidum (inoculated on 18th of September 2014)
PCO1 _ pleurotus cornucopie (inoculated on 18th of September 2014)
Armillaria Mellea after 6 days, 9 days and 21 days:
Theo Spyropoulos 01
“The architecture itself is not designed to a site in a conventional sense but we have to try at least […] to think about certain fitness criteria that will allow the system to develop possibilities, different sites and configurations.”
– Theo Spyropoulos
John Frazer 01
“The aim of an evolutionary architecture is to achieve in the built environment the symbiotic behavior and metabolic balance that are characteristic of the natural environment.”
– John Frazer
Phil Ross 01
“By providing enviroments to live within, we can appriciate the details of a framed existence. Our attention is drawn not only to isolated living element, but also to the greater organization from which it has been removed. The small parts, once enclosed and removed from their native residence, are where we truly focus our gaze, their isolation serving as a lens through which to imagine an entire living world.”
– Philip Ross































































Devi effettuare l'accesso per postare un commento.